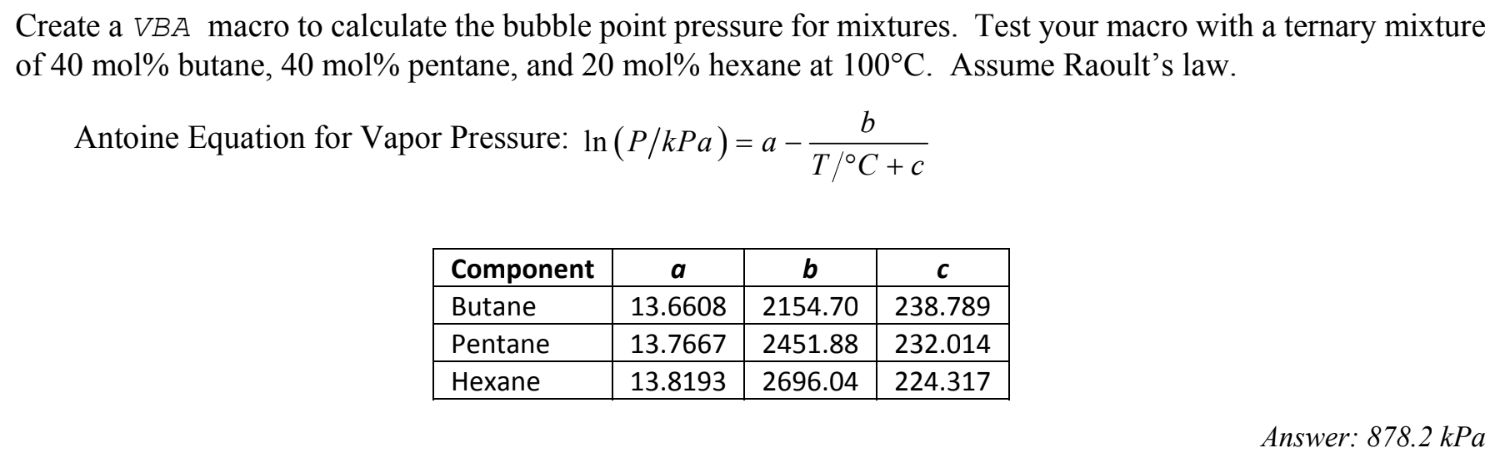
Computational algorithm based on bubble point pressure calculations for... | Download Scientific Diagram

DOC) Calculating Bubble & Dew Points for Ideal Mixtures P A -Partial pressure of component A P o A -Vapour pressure of component A xA -Liquid mole fraction of component A
Introduction to Chemical Engineering Processes/Vapor-Liquid equilibrium - Wikibooks, open books for an open world

SOLVED: Problem 6.60 Bubble and Dew Points from Raoult'Law Calculate the following: The bubble point temperature of liquid mixture with molar composition of 70.0 mol% n-hexane and 30.0 mol% n-heptane at 1.00
![SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat](https://cdn.numerade.com/ask_images/1604588eda0f4ffc8897da2c6b3d4fee.jpg)
SOLVED: 5) [40 pt] In nonideal mixtures bubble point temperature calculations can be performed using modified Raoult s law (y P-XirPra ) by finding such that the following equation is satisfied: XiYaPsat